Spotlight on a Researcher – Dr. Travis Craddock

 In the past decade digital transformation, powered by the latest innovations in artificial intelligence (AI) and machine learning, has influenced almost all aspects of the drug development process from screening therapeutic targets to management of clinical trial data. This innovative field, with a multitude of potential applications, has attracted the attention of scientists in various universities and research institutes. Travis Craddock, Ph.D., the Director of the Clinical Systems Biology Group at Nova Southeastern University’s (NSU) Institute for Neuro Immune Medicine (INIM; KPCOM), is one of them.
In the past decade digital transformation, powered by the latest innovations in artificial intelligence (AI) and machine learning, has influenced almost all aspects of the drug development process from screening therapeutic targets to management of clinical trial data. This innovative field, with a multitude of potential applications, has attracted the attention of scientists in various universities and research institutes. Travis Craddock, Ph.D., the Director of the Clinical Systems Biology Group at Nova Southeastern University’s (NSU) Institute for Neuro Immune Medicine (INIM; KPCOM), is one of them.
Dr. Craddock and other members of his lab work to identify ideal drug candidates and understand potential interactions of therapeutic molecules with biological entities other than the intended target in the body. These considerations are essential for developing efficient and safe therapeutic intervention strategies for various diseases. Identifying appropriate targets is especially challenging in the case of complex diseases, which cannot be attributed to a single underlying cause and where the traditional concept of “one disease, one drug” does not apply. These complex diseases result from a combination of environmental, genetic, and lifestyle factors. Owing to this complex etiology, diagnosis and treatment of diseases such as Gulf War Illness (GWI) and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) is even more difficult.
To overcome these challenges Dr. Craddock is working on developing a computational pipeline that implements AI and machine learning to identify drug target candidates for possible treatment of complex diseases like the above, and to design better therapeutic strategies for such diseases.
In a peer-reviewed research article published in the International Journal of Molecular Sciences in 2018, researchers in Dr. Craddock’s lab at NSU and collaborators from other universities reported how drug docking programs (listed next) can be implemented to assess off-target interactions of therapeutic compounds (i.e., drug side effects). In this publication the researchers applied AutoDock 4.2, AutoDock Vina, and Schrodinger’s Glide to determine non-specific immune and hormonal interactions of 43 FDA-approved drugs used for treating GWI symptoms. This research indicates that many of these approved drugs bind to multiple hormonal and immune targets in the body, which may cause negative side effects in a patient, implying that their use in the treatment of GWI should be done with caution. Studies like these enable researchers to identify and eliminate medicinal compounds with possible harmful off-target effects early in the drug development process.
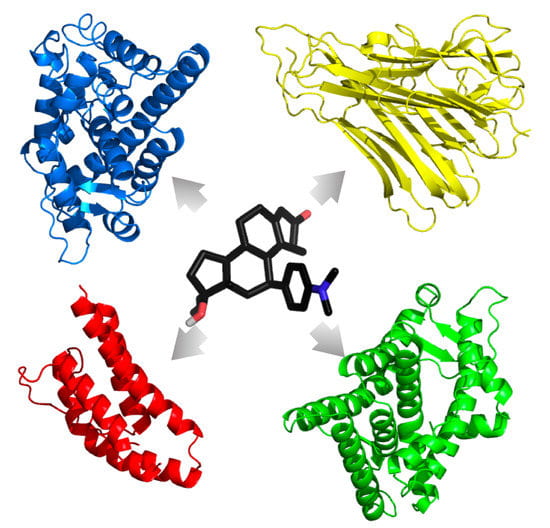
Visualization of the side effects of a drug molecule.
Another line of work from Dr. Craddock’s lab, which has broader implications, is the development of a meta-database for mapping interactions between drugs, genes, and proteins to facilitate identification of potential adverse drug events for treatments involving multiple drugs. Functioning of this meta-database named DRUGPATH, and how it can contribute to designing the ideal treatment regimen, was published in the International Journal of Molecular Sciences in 2020. As reported in this peer-reviewed article, DRUGPATH contains information on 12,940 unique drugs, 3,933 unique pathways, 5,185 unique targets, and 3,662 unique genes. It uses reliable resources such as the Pharmacogenomics Knowledgebase (PharmGKB), DrugBank, the HUGO Gene Nomenclature Committee (HGNC), the Guide to Pharmacology (GTP), and the FDA’s National Drug Code Database (FDA NDC) to determine 59,561 unique drug-gene interactions, 77,808 unique gene-pathway interactions, and over 1 million unique drug-pathway interactions. To validate the efficacy of this database, researchers at NSU evaluated two drug combinations, the findings of which were published in the above research article.
This meta-database is supported by INIM and available online through for use by other researchers. The comprehensive mapping of possible interactions between drugs, genes, targets, and biological pathways offered by the DRUGPATH meta-database will enable clinicians to be aware of potential adverse effects of drugs. This will help them predict and prevent possible negative side effects resulting from the combination of drugs or their interactions with biological targets.

Clinical Systems Biology Group members.
One of the key areas of focus for Dr. Craddock’s research is GWI. This complex disease involves neuroinflammatory responses, alteration of regulatory pathways, and immunological dysfunction. The behavioral and physiological implications of this illness are complex and believed to be associated with external stressors, altering of the permeability of the semipermeable blood-brain-barrier (BBB), and signaling involving mediators such as cytokines, chemokines, and hormones.
To better understand this complex regulatory system, Dr. Craddock developed a logic model representation of the neuroimmune system across the BBB connecting the CNS to the peripheral immune system. The findings of this research by Dr. Craddock and colleagues on neuroimmune interactions in Gulf War Illness (GWI) was published in 2021 in the International Journal of Molecular Sciences. The researchers compared clinical samples of GWI patients (with and without PTSD symptoms) to mouse models of GWI in blood and used the corresponding neuroimmune states in the mouse brain to infer the state of the BBB in GWI subjects. The data obtained from this comparative analysis offer a better understanding of the signaling pathways and alterations in the regulation of the neuroimmune system in these patients. These findings will enable researchers to develop predictive models for treatment that have the potential to improve regulation of the neuroimmune system of individuals suffering from GWI.
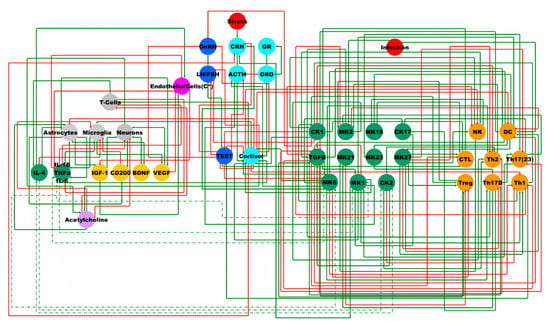
Neuroimmune signaling network, a model for a complex disease.
In addition to resulting in multiple academic publications, research being conducted at Dr. Craddock’s lab at NSU’s INIM has substantial translational and commercial potential. For example, Dr. Craddock developed a new technology for predicting possible clinical interventions to treat multifactorial diseases. This invention can be used to predict possible therapeutic strategies for complex diseases and determining likely off-target deleterious effects of drug candidates. Following his discovery Dr. Craddock worked extensively with NSU’s Office of Technology Transfer to file a patent application for this innovation. The proposed technology invented by Dr. Travis Craddock and his collaborator, Dr. Gordon Broderick employs AI and machine learning for more accurate prediction of clinical intervention impacts for complex and multifactorial diseases. This invention will allow researchers to construct regulatory networks based on protein-protein interaction networks using a computer, and it will enable them to conduct drug target selection and screening much quicker than traditional screening methods.
The ability of this computational platform to identify drug targets by cross referencing with known pharmacogenomics databases makes it the ideal tool for selecting drug candidates for a multi-drug, multi-target-based therapy. Dr. Craddock and other researchers at NSU have applied this innovation with success to identify interactions between therapeutic compounds used to treat GWI as well as between other drugs used for treating comorbidities such as hypertension and hyperlipidemia.
As the Director of Clinical Systems Biology Group at INIM, Dr. Craddock is leading the efforts in NSU’s emerging expertise in the use of machine learning and AI for developing medical therapies. The research being done by Dr. Craddock’s lab will result in development of novel methods for treating complex diseases such as GWI and ME/CFS. Unlike more conventional drug target selection and screening technologies, the machine learning-based method invented by Dr. Craddock can identify potential interactions with non-target entities and thus reduce the chances of negative side effects resulting from therapeutic molecules. This will significantly reduce time and improve the efficacy of selecting drug candidates for complex diseases.
Dr. Craddock has made valuable contributions to the field of machine learning for drug development, and in the future his research work at NSU’s INIM will lead to further innovations in the treatment of complex diseases. These scientific findings will further advance pharmaceutical research as well as improve patient outcomes for those suffering from GWI and other complex diseases.